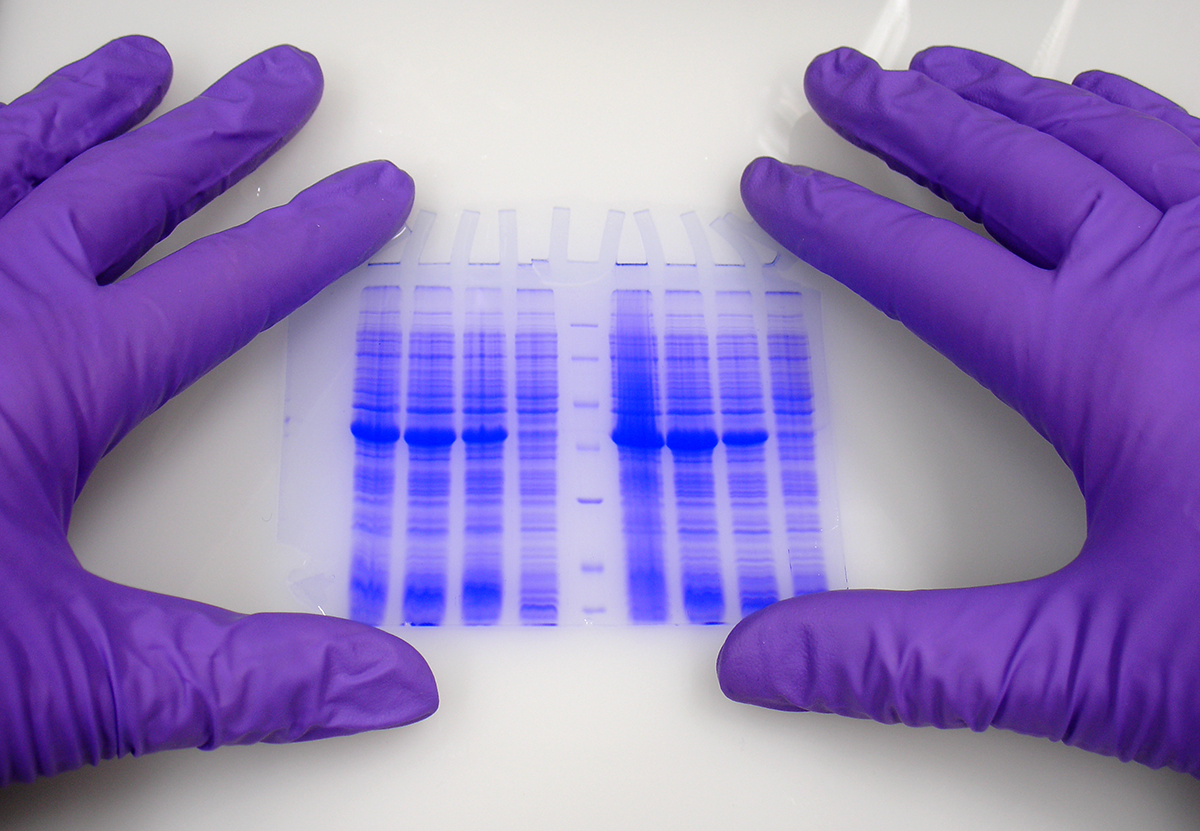
ARVYS Proteins Inc. provides a full spectrum of protein biochemistry services – recombinant protein expression in bacterial, insect and mammalian cells, protein purification, refolding, assays and assay development, protein characterization, fermentation and endotoxin removal.
Outsource your protein biochemistry projects to ARVYS and enjoy superior results, team expertise and customer support after project completion.
Today we look at the publication from Humana Press. Edited by Valentin Köhler Department of Chemistry, University of Basel, Switzerland
Preface
The second edition of protein design in the Methods in Molecular Biology series aims at providing the reader with practical guidance and general ideas on how to approach a potential protein design project. Considering the complexity of the subject and its attention in the scientifi c community it is apparent that only a selection of subjects, approaches, methods, studies, and ideas can be presented. The design of well-folded peptidestructures and the redesign of existing proteins serve multiple purposes from potentially unlimited and only just developing applications in medicine, material science, catalysis, the realization of systems chemistry, and synthetic biology to a deeper understanding of molecular evolution. The book is roughly organized in increasing complexity of the systems studied. Additional emphasis is put on metals as structure-forming elements and functional sites of proteins towards the end. A computational algorithm for the design of stable alpha-helices is discussed in the first chapter and is accessible in the form of a web-based tool. An extensive review on monomeric β-hairpin and β-sheet peptides follow. In the design of these species, any tendency to self-assemble has to be carefully considered. In contrast, Chapter 3exploits just this phenomenon—peptides engineered to self-assemble into fibrils. Subsequently, some possibilities and aspects resulting from the incorporation of unnatural amino acids are outlined. In the practical methods chapter on the redesign of RNase A, a variable α-helical fragment is reassembled with the remainder of the protein structure, generated by enzymatic cleavage. Chapter 5discusses the design and characterization of fluorinated proteins, which are entirely synthetic. Comparisons to non-fluorinated analogous structures are included and practical advice is offered. This is followed by an overview of considerations for the generation of binary- patterned protein libraries leading on to library-scale computational protein design for the engineering of improved protein variants. The latter is exemplified for cellobiohydrolase II and a study aimed at changing the co-substrate specificity of a ketol-acid reductoisomerase. Chapter 8focuses on the elaboration of symmetric protein folds in an approach termed “top-down symmetric deconstruction,” which prepares the folds for subsequent functional design studies. The identification of a suitable scaffold for design purposes by means of the scaffold search program ScaffoldSelection is the topic of Chapter 9 . The computational design of novel enzymes without cofactor is demonstrated for a Diels-Alderase in Chapter 10 . The final four chapters deal with metal involvement in the designed or redesigned structures, either as structural elements or functional centers. The begin is made with a tutorial review that imparts general knowledge for the design of peptide scaffolds as novel pre-organized ligands for metal-ion coordination and then exemplifies these further in a respective case study. This is followed by an introduction on the computational design of metalloproteins, which encompasses metal incorporation into existing folds, fold design by
Abstract
Recent studies have elucidated key principles governing folding and stability of α-helices in short peptides and globular proteins. In this chapter we review briefly those principles and describe a protocol for the de novo design of highly stable α-helixes using the SEQOPT algorithm. This algorithm is based on AGADIR, the statistical mechanical theory for helix-coil transitions in monomeric peptides, and the tunneling algorithm for global sequence optimization.
Conclusions:
In this chapter we have presented the SEQOPT method for the rational design of α-helices based on proteinogenic amino acids, to achieve a high conformational stability by global optimization of the protein segment/peptide sequence. The method has three key characteristic properties: (1) only the 20 standard amino acids can be used, (2) it offers the possibility to arbitrarily fix any functionally important fragments of the primary structure, and (3) it offers accordingly the possibility to optimize the helical content of only those fragments that do not contain important functional groups of the protein. It has been shown that the proposed method is an effective tool for protein engineering [56]. In contrast to other methods for global energy optimization (molecular dynamics, Monte Carlo, etc.) that are often used to engineer the stability of the protein under investigation by altering only one or two amino acid residues and searching for advantageous physical interactions, the SEQOPT method deals with all possible sequences of protein α-helices and selects a suitable solution for most practical purposes.
To buy the full journal, click here.
ARVYS Proteins Inc. provides a full spectrum of protein services to the life science, pharmaceutical and biotechnology communities. Our work experience encompasses almost every aspect of protein biochemistry allowing us to contribute to projects regardless of whether they are at early research or late development stages. We can be your partner in:
– generation and expression of recombinant proteins in bacterial, baculovirus and mammalian expression systems,
– large-scale fermentation,
– cell culture,
– purification of recombinant proteins, antibodies or naturally occurring proteins,
– refolding from inclusion bodies,
– improvement of protein stability,
– protein labeling with fluorescent, biotin and enzyme probes,
– endotoxin removal and testing for in vivo studies,
– protein characterization to monitor its integrity and functionality
“Don’t think clean thoughts with dirty enzymes” – is a famous saying of Efraim Racker, one of the founding fathers of modern biochemistry. We believe this philosophy should be extended to all proteins – purified and well-characterized preparations will save you time, effort and resources. Your confidence in protein reagents is the core focus of our business.